Which Best Describes a Hypertonic Solution
The plasma membrane. C Water will move out of the cell.
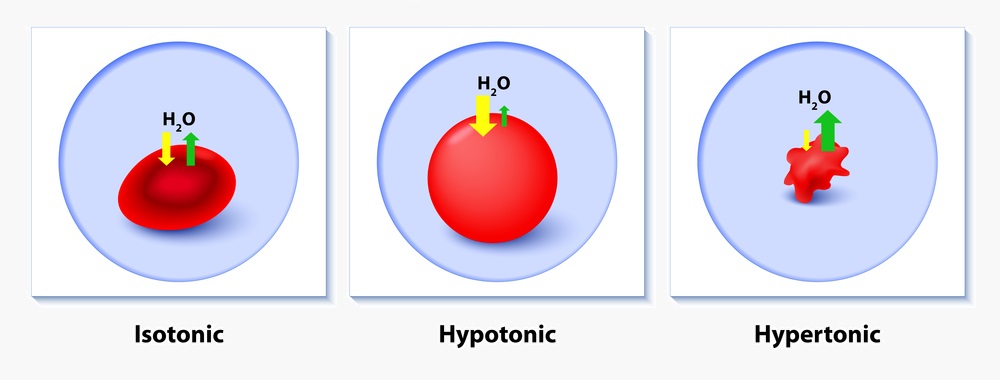
Hypertonic Solution Definition And Examples Biology Dictionary
The solvent is the substance that.
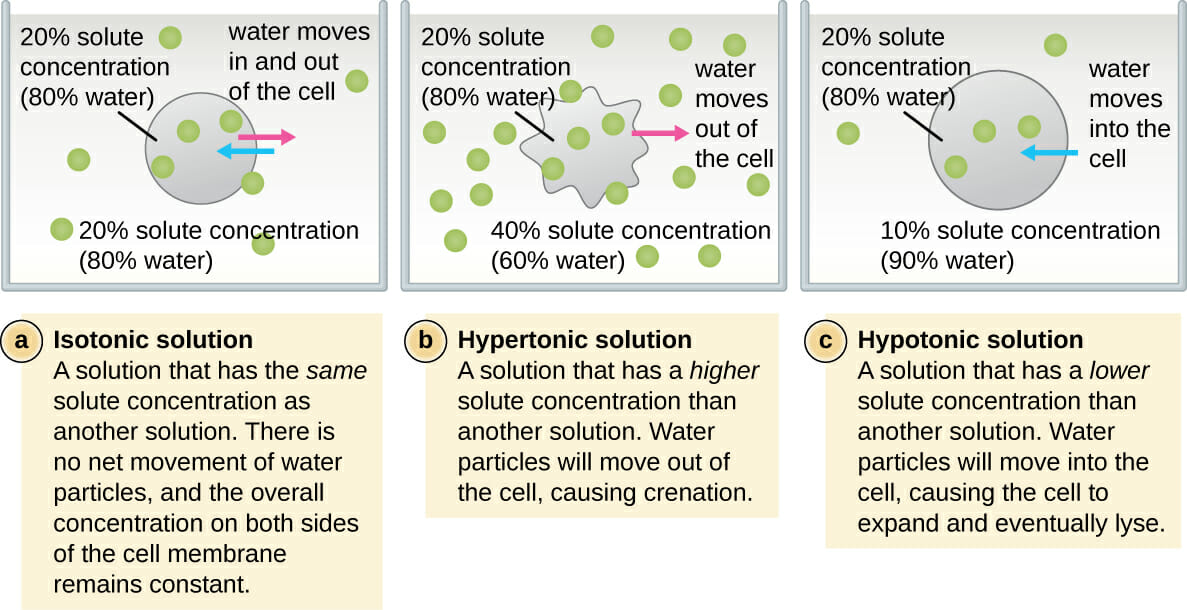
. What are the solutions to the equation 2x218 the possible answers are A. Giving hypertonic solutions can cause a risk for hypernatremia and volume. A cell is placed in a hypertonic solution.
This causes water to rush out making the cell wrinkle or shrivel. Chose the term or phrase that best describes how exocytosis functions. When a cell is placed in a hypotonic environment water will enter the cell and the cell will swell.
Hypertonic blood draws water out of the interstitial fluid which makes the interstitial fluid hypotonio. A hypertonic solution is one which has a higher solute concentration than another solution. Contains more oxygen crystals than the body plasma.
Scientists must describe cell contents compared to the environment. This in turn makes the interstitial fluid lose water to the colls. 3 Which of the following statements best describes what happens when a bacterial cell is placed in a solution containing 5 NaCl.
The opposite solution with a lower concentration is known as the hypotonic solution. G1 occurs first followed by DNA replication in the S phase followed by G2. Which statement correctly describes a hypertonic solution when compared to another solution.
2 solutions -9 B. The outside solution has higher soluble concentration than inside the cell. Chemistry questions and answers.
Hypertonic solutions have a higher solute concentration than inside the cell. Asked Oct 25 2021 in Biology Microbiology by Rioux. Which of the following best describes a cells response to being placed into an extracellular solution that is hypertonic to the cytosol.
If a cell is placed in a hypertonic solution water will leave the cell and the cell will shrink. This in turn causes water to move into the cells. The solute particle concentration in the solution is less than half of the solute particle concentration inside a living cell.
Contains less electrolytes than the body plasma D. Which of the following best describes a hypertonic solution. Which of the following below best illustrates the relationship between the comparison of the 2 following terms.
Concentration higher than the body plasma B. Hypertonic solutions are solutions that have a greater concentration of solute compared to the cell. A Water will move into the bag causing it to swell.
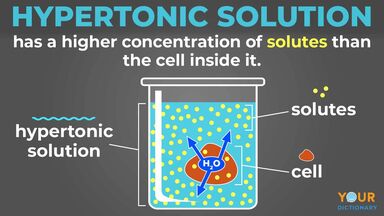
Hypertonic Solution Definition. A hypertonic solution contains a higher concentration of solutes compared to another solution. Which scenario will most likely happen.
An example of a hypertonic solution is the interior of a red blood cell compared with the solute concentration of fresh water. If a cell is placed in a hypertonic solution the cell will shrink due to water osmotically moving out. Locate a region of healthy cells and sketch a few adjacent cells.
Similar stalks left in a 015 M salt solution become limp. The solution is more concentrated with solutes than the other solution. While touching one corner of the coverslip with a piece of Kimwipe to draw off the.
Question 5 1 point Select which answer best describes a hypertonic solution. Which of the following best describes what will happen after one hour. Water will move into the cell and cause it to swell or burst.
Hypertonic solutions have a higher solute concentration. 2 solutions -3 D. Asked Jul 1 2020 in Chemistry by Gibbz.
The solute particle concentration in the solution is equal to the solute particle concentration inside a living cell. If a cell is placed in a hypertonic solution the cell is considered. This is clearly seen in red blood cells undergoing a process called crenation.
A and the salt solution are both hypertonic to the cells of the celery stalks B and the salt solution are both hypotonic to the cells of the celery stalks C is hypotonic and the salt solution is. Water add a drop of concentrated salt solution to. Celery stalks that are immersed in freshwater for several hours become stiff.
D Water will move into the cell. Hypertonic solutions are volume expanders. Which most accurately describes the phases contained within interphase.
Read each of the following procedural steps for plasmolyzing Elodea sprig and then select the correct order below. A Sucrose will move into the cell from a higher to a lower concentration. B The cell will undergo osmotic lysis.
A hypertonic solution is one that has a higher solute concentration outside the cell than inside. If a cell is placed in a hypertonic solution what will happen to the cell. The saline that is hypertonic describes any preparation of saline that has over a concentration of 09 of sodium chloride which is the normal saline.
Some of the example that support these to be a good example of hypertonic solution example are-3 and 2 Hypertonic saline. Causes a cell to shrink. Compare hypertonic hypotonic and isotonic solutions.
2 solutions -3 C. Thus water molecules move from inside to outside the cell. Which of the following best describes hypertonic solution.
All of the following are a level of organization in the human body except which one. From this we can deduce that the freshwater _____. Hypertonic solutions assist in restoring the circulating volume by bringing the water out of the intracellular space causing the extracellular fluid volume to increase.
Concentration lower than the body plasma C. Tonicity in living systems. Putting your trash out of the house.
In an isotonic environment there is no net water movement so there is no change in the size of the cell. Hypertonic blood will lose water to the interstitial fluid which makes the interstitial luid hypertonic. Sketch the plasmolyzed cell.
The solvent is the substance that dissolves the solute. Plant cells in a hypertonic solution can look like a pincushion because of whats going on inside. Question 18 of 33 A student fills a semipermeable bag with 0100 M aqueous glucose solution and places it in a hypertonic aqueous solution of 0800 M glucose.
I am completely and utterly lost so if someone could help me just like figure.
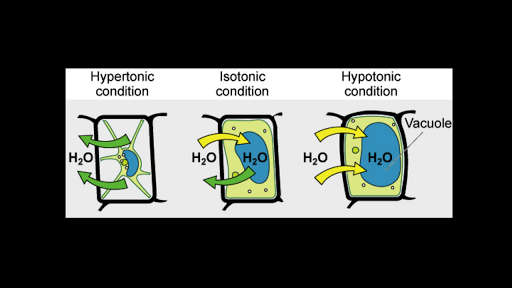
Tonicity Hypertonic Isotonic Hypotonic Solutions Article Khan Academy

Isotonic Vs Hypotonic Vs Hypertonic Solution Biology
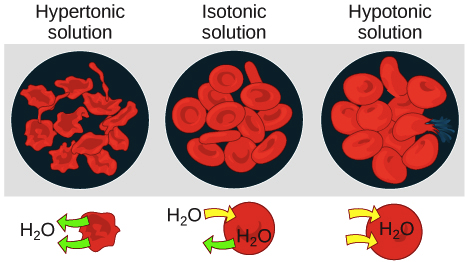
Tonicity Hypertonic Isotonic Hypotonic Solutions Article Khan Academy

Comments
Post a Comment